
electric wheelchair
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request detailsUse the dynamic catalog below to request documentation for hospital beds, smart patient-room platforms, rehabilitation equipment, remote monitoring tools, and selected surgical support devices.

Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details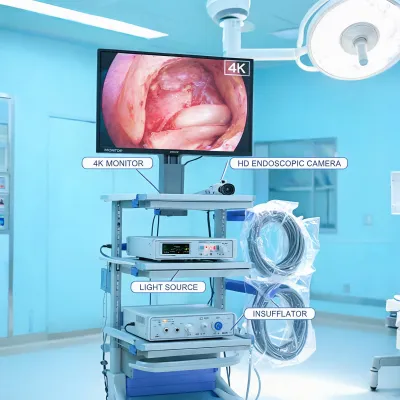
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details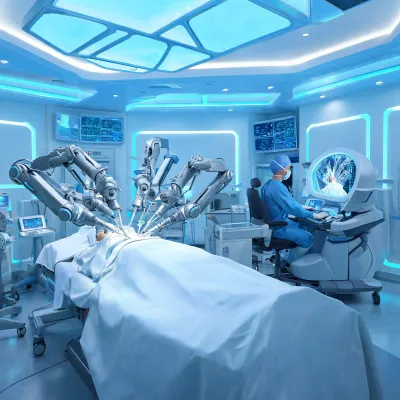
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details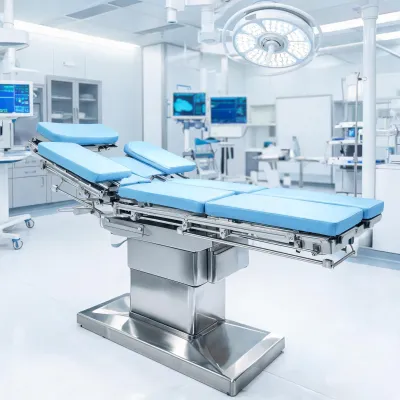
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request details
Request IFU references, service tier assumptions, caregiver training scope, connectivity notes, and procurement documentation for this equipment family.
Request detailsHill Rom product reviews usually involve several groups at once: nursing leadership checks workflow fit, biomedical engineering checks electrical safety and maintenance assumptions, infection prevention checks cleaning and surface management, IT checks connected-device requirements, and finance checks acquisition model versus long-term service cost. The product grid is therefore intentionally inquiry-led. A buyer can open a request from any product family and ask for the matching IFU, preventive maintenance schedule, accessory compatibility list, warranty scope, spare-parts assumptions, training plan, and market-specific regulatory documentation.
For hospital bed and patient-room programs, the practical comparison is rarely limited to purchase price. Reviewers also need fleet age, room mix, fall-risk protocol, caregiver staffing model, nurse call architecture, cleaning turnover target, replacement parts availability, and whether the facility expects direct purchase, lease, rental, or refurbished equipment. Hill Rom can package these assumptions into a value analysis file so each stakeholder evaluates the same operating model rather than separate catalog fragments.
When a product request involves connected assets, the review can also include cybersecurity documentation, interface assumptions, user-access controls, alarm-routing expectations, and software update responsibilities. These details help IT and clinical engineering decide whether a product family can enter an existing patient-room technology stack without creating avoidable operational risk.